Abstract
Background: Cerebral vasculopathy is a frequent complication of sickle cell anemia (SCA) and is associated with a high risk for stroke. This vasculopathy seen in SCA can be progressive and severe. Sickle cell patients with severe vasculopathy, including Moyamoya syndrome are at increased risk for neurological disabilities and death. While chronic transfusions decrease the risk of stroke in SCA; unfortunately, progression of vasculopathy can occur despite treatment. Limited data exists regarding long term outcomes for this population. We evaluated effectiveness of three treatment approaches at our center, namely chronic transfusions, surgical revascularization plus chronic transfusions and allogenic hematopoietic stem cell transplant (HSCT).
Methods: A retrospective chart review was preformed to identify patients with SCA (hemoglobin SS, Sβ0) and severe vasculopathy including Moyamoya syndrome between 1986 to 2017. Severe vasculopathy was defined as having at least one cerebral artery with > 70% stenosis and/or occlusion as seen on MR angiogram (MRA), CT angiogram (CTA) or conventional angiogram (DSA) as determined by a neuroradiologist at our institution. Patients were identified from an institutional stroke database. Patients were included for analysis if they received at least one of the following: chronic transfusions, surgical revascularization (i.e. encephalo-duro-arterio-synagiosis (EDAS) plus chronic transfusions or HSCT. For HSCT, all graft types (bone marrow, peripheral blood stem cells, umbilical cord blood), conditioning regimens and donor types (related, unrelated and haploidentical) were included. Time to event analyses were performed from the time of intervention (transfusion, HSCT, EDAS/chronic transfusions) using overt clinical stroke, new silent infarcts, progression of vasculopathy or new vasculopathy. Survival curves were analyzed using the log-rank (Mantel-Cox) test.
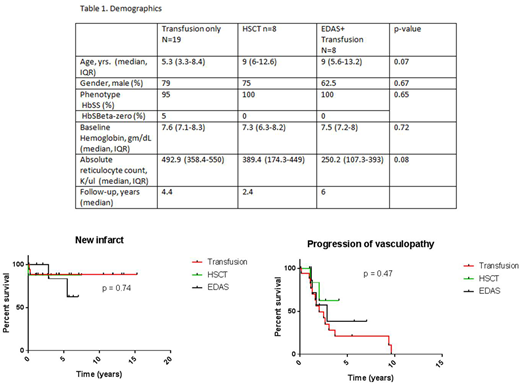
Results: Of 35 patients identified, 54% (n =19) underwent chronic transfusions, 23% (n=8) of patients underwent HSCT after being on chronic transfusions, 23% (n=8) underwent EDAS with chronic transfusions and 1 patient underwent each of the above three modalities (Table 1). Median age at time of intervention was similar for all three cohorts (Table 1). Males were overrepresented in all treatment arms (62.5-79% of patients). Average hemoglobin level prior to intervention was also similar: 7.6 g/dL (IQR 7.1-8.3) for the chronic transfusion cohort, 7.3 gm/dL (IQR 6.3-8.2) for the HSCT cohort, and 7.5 gm/dL (IQR 7.2-8) for the EDAS/chronic transfusion cohort. Absolute reticulocyte count was 492.9 K/ul (IQR 358.4-550) for the chronic transfusion group, 389.4 (IQR 174.3-449) for HSCT, and 250.2 (IQR 107.3-393) for EDAS/chronic transfusions (p=0.08). One patient died of overt stroke in the chronic transfusion cohort. The median follow-up times for the transfusion, HSCT and EDAS plus transfusion groups were 4.4, 2.4 and 6 years respectively.
Time from date of intervention (transfusion, HSCT, EDAS) to overt clinical or silent stroke was evaluated (Fig 1). Two of the nineteen patients in the chronic transfusion cohort suffered an overt stroke, while one of eight and two of eight had strokes in the post-HSCT and EDAS plus chronic transfusion cohorts respectively. Fourteen of nineteen (74%) in the chronic transfusion cohort had progression of severe vasculopathy after being on transfusions while two of eight (25%) in the HSCT and four of the eight (50%) patients in the EDAS plus chronic transfusion cohorts had progression. The one patient with all three different interventions did not have additional infarction (clinical or silent) or vasculopathy progression during 1.5 years of follow-up.
Conclusions: The risk for cerebral infarction and/or vasculopathy progression after initiation of treatment with either chronic transfusion, HSCT or EDAS is still a major concern. Our data suggest HSCT and surgical revascularization with chronic transfusion provide the greatest benefit in reducing stroke risk and HSCT reduces risk for progression of a severe vasculopathy. Additional, large population studies are needed to clarify the risk.
Majumdar:NIMHD: Research Funding. Campbell:Functional Fluitics: Membership on an entity's Board of Directors or advisory committees; Global Blood Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal